MGI is bringing its full genomics expertise and resources to the front lines in the fight against the 2019 novel coronavirus.
MGI has scaled up production of its new real-time fluorescent RT-PCR kits for detecting the virus, known as 2019-nCoV, donating 20,000 kits to support the response in Wuhan and Hubei Province. The test can issue results in a few hours. MGI has now released about than 50,000 test kits to hospitals and disease control centers around China.
MGI Chairman and Co-Founder Wang Jian and a team of employees arrived in Wuhan January 25 to lead MGI's emergency response. During the Chinese New Year holiday, the company set up an emergency coordination center in Wuhan to mobilize efforts and expand the manufacturing capacity, with staff working around the clock to produce the testing kits and arrange storage, transportation and other logistics. MGI said its teams from Wuhan, Shenzhen, Tianjin and other cities where it has offices or laboratories are on standby to provide the fastest response to frontline efforts.
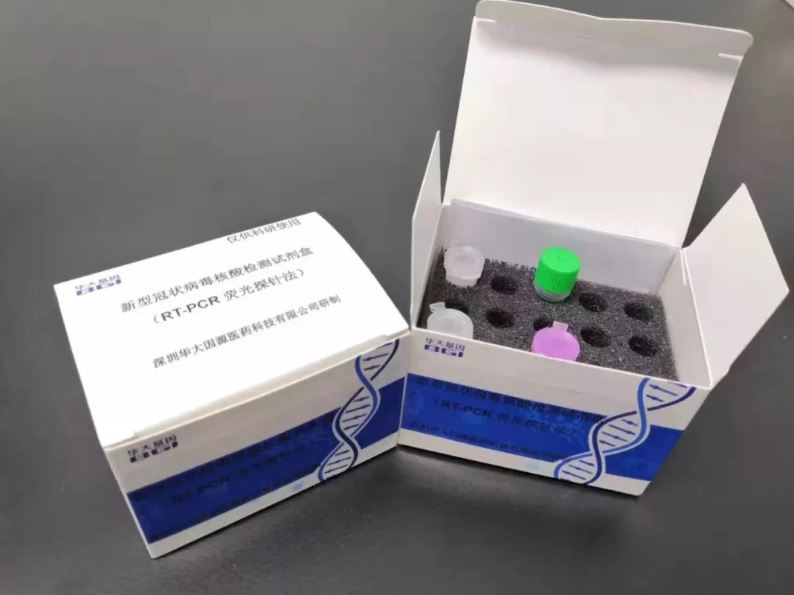
In addition to the rapid detection kit based on RT-PCR technology, MGI has also developed a metagenomic sequencing detection kit (2019-nCoV nucleic acid detection kit-combinatorial Probe-Anchor Synthesis sequencing method) tomonitor mutations. The metagenomic sequencing kit canidentify and diagnose other coronaviruses, including new coronaviruses and respiratory tract infections, and achieve rapid detection of viral sequences. The combination of RT-PCR and MGI metagenomics solution PMseq® can detect both known and novel microorganisms, in addition to 2019-nCoV. The two coronavirus detection kits and the ultra-high-throughput DNBSEQ-T7 sequencing system and analysis software have officially passed the emergency approval procedures of the National Medical Products Administration (China's FDA), becoming the first batch of officially tested products to fight the outbreak. Two of the powerful DNBSEQ-T7 sequencers, produced by MGI, have been deployed to Wuhan.

MGI has been working with various Chinese authorities, including the Chinese Center for Disease Control and Prevention, to deliver detection kits to various hospitals and local disease control centers. MGI is the third-party collaborator authorized by China’s National Health Commission in detecting the 2019-nCoV virus in China. Further, MGI is also engaged with relevant organizations overseas to supply the test kits and offer additional pathogen detection solutions using next generation sequencing technology. International delivery has started, as the kits are equipped with English manuals and offer online training programs. On January 24, a Chinese citizen in Brunei, suspected of 2019-nCoV infection, was diagnosed negative as the first international case using the MGI kit.


 Sequencer Products: SEQ ALL
Sequencer Products: SEQ ALL













 Technologies
Technologies Applications
Applications Online Resources
Online Resources Data Bulletins
Data Bulletins Service & Support
Service & Support Global Programs
Global Programs Introduction
Introduction Newsroom
Newsroom Doing Business With Us
Doing Business With Us Creative Club
Creative Club













